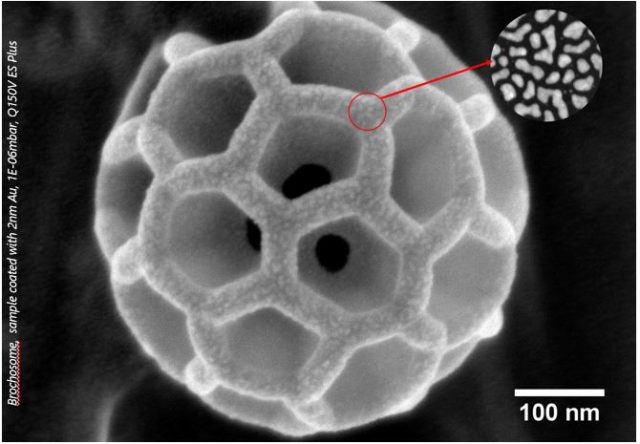
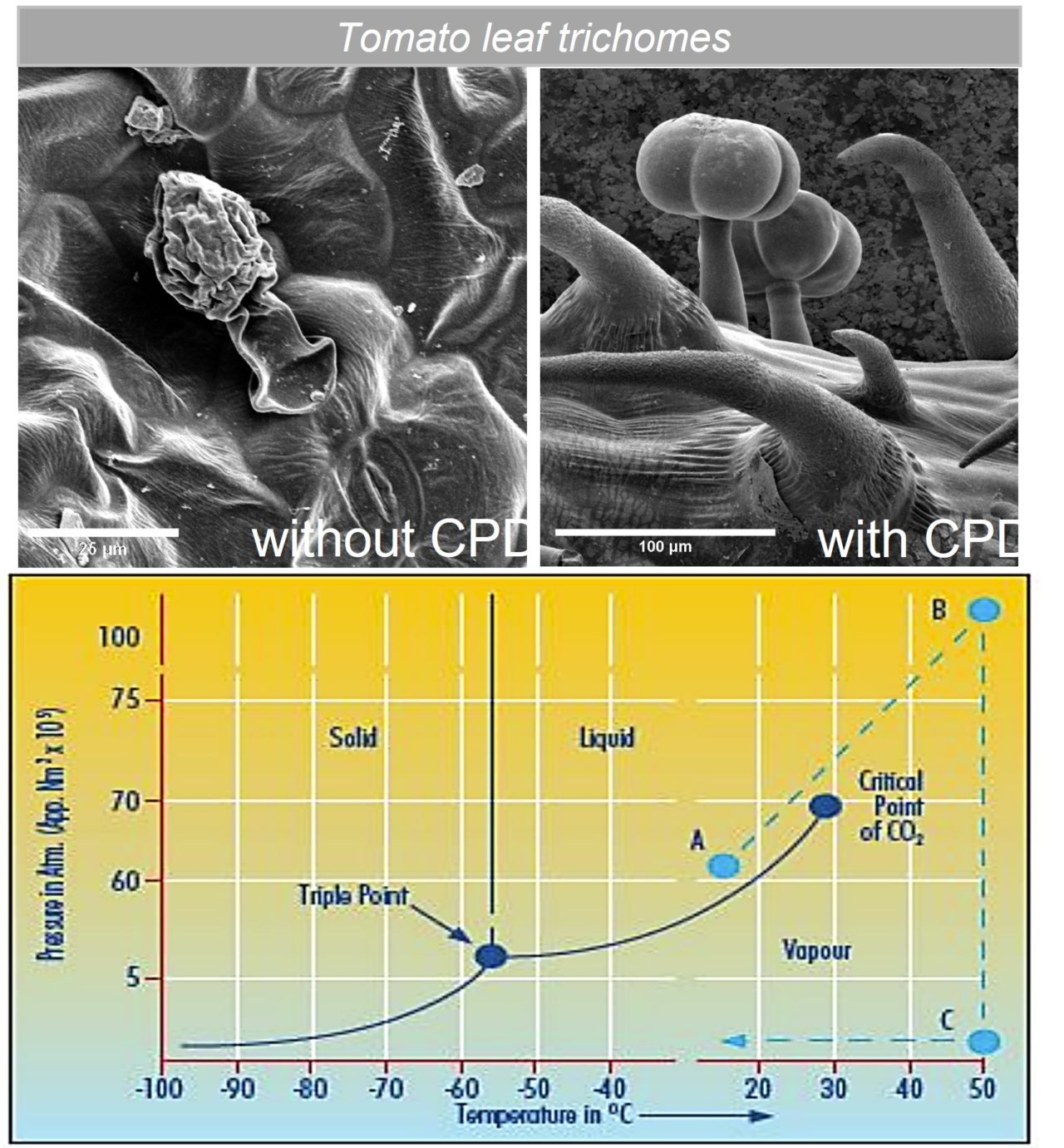
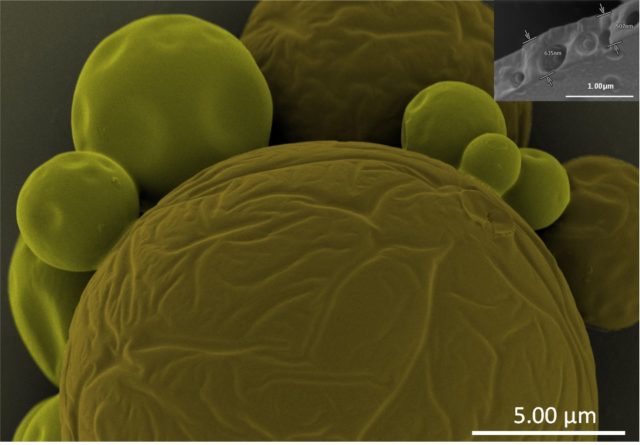
Electron microscopy techniques rely on the transfer of electrons between sample and microscope. For conductive samples, this is easily achieved – however, non-conductive, or poorly conducting samples must be coated with an electrically conductive coating to produce usable images. A high-quality coating is essential to obtain high-quality images. Quorum Technologies developed the Q Plus Series to provide researchers with a versatile and high-performance coating to rival major manufacturers, without the associated price tag.
The Role of Coatings in Electron Microscopy
Scanning Electron Microscopes (SEM) and Transmission Electron Microscopes (TEM) work similarly to Optical Microscopes but rather than probing materials using light, they use electrons. Optical microscopes are diffraction-limited (to a maximum resolution of about 200 nm) whereas electron microscopes can produce beams of electrons with much smaller wavelenght1 and surpass the resolving power of optical microscopes by several orders of magnitude. As a result, they are the most powerful microscopy techniques in the world.
Using electrons instead of light, however, introduces other complications. Both techniques (SEM and TEM) rely on the transfer of electrons between sample and microscope; therefore, it can be difficult (or near impossible) to obtain a usable image signal from samples with poor or no conductivity. This is especially true of SEM, where samples are bombarded with an electron beam: poorly conductive or non-conductive samples will rapidly accumulate charge under these conditions, leading to image distortion as well as thermal and radiation damage to the sample. In extreme cases, the sample may accumulate sufficient charge to decelerate the primary beam, acting as an “electron mirror” and preventing an image altogether.2
To overcome this, poorly conducting samples are coated with a thin layer of metal or carbon. This makes the surface conductive, eliminating charge accumulation and enabling a better signal to be obtained by the microscope. Coating techniques are widely used for imaging biological or organic samples since these are typically non-conductive and easily damaged by the electron beam.
While the primary role of coating in SEM is to increase electrical conductivity and prevent “charging”, it also has several other useful effects:
- Coating a sample with a thermally conductive material such as gold, silver, copper, aluminium can reduce thermal damage from the primary electron beam.
- Particulate matter and fragile organic samples can be mechanically stabilized and held in place by a thin layer of carbon.
- Coating organic samples that contain trapped gas or moisture protects both sample and microscope from being contaminated by off-gassing.
- Metallic coatings can be used to minimize the volume of penetration of the electron beam, localizing scanning to the very surface of a sample. This can also dramatically increase the emission of secondary and backscattered electrons.
Download our Guide to Coating for Electron Microscopy Here
The Impact of Coating Quality
When working with a coated sample in an electron microscope, it is the coating itself that gets directly imaged. The quality of the coating, therefore, places a hard limit on the quality of the images that can be obtained.
When imaging very small structures (such as electrospinning fibres doped with copper nanocrystals), depositing a coating that is too thick can easily bury meaningful information. It is vital that coating thickness can be precisely controlled and tailored to the features that are being interrogated.3
In the worst cases, poor quality coating equipment introduces contamination and can irreparably damage samples. Researchers often opt for cheap coaters to save money, only to find that their costs increase due to additional microscope time and ruined samples.
However, thanks to the Q Plus Series from Quorum Technologies, it is no longer necessary to pay a premium to obtain state-of-the-art coatings.
The Q Plus Series: Affordable and High-Quality Coating
The Q Plus Series is the latest iteration of Quorum’s world-leading range of coaters; offering cutting-edge sputter and evaporation coating in a single easy-to-use platform. Quorum’s turbomolecular-pumped coaters are suitable for both oxidizing and non-oxidizing metals, while our low-cost rotary-pumped sputter coaters are suitable for non-oxidizing metals. The Q Plus Series is suitable for sputter coating and evaporating carbon coating for SEM, FE-SEM and TEM applications.
This new range of coaters is designed to enable researchers to exercise precise control over coating thickness, whatever their application requirements. For the highest level of performance, the Q150V Plus provides an ultimate vacuum of 10-6 mbar; removing oxygen, nitrogen and water vapour from the chamber and eliminating chemical reactions during the sputtering process. The Q150V Plus also enables the production of finer grain size and thinner coating for ultra-high-resolution applications (beyond 200,000x magnification). Low scattering enables the formation of high-purity amorphous carbon films of high density.
All models in the Q Plus Series feature a touch-screen interface as well as status LEDs and audio notifications for straightforward and intuitive control. Integrated 16 GB memory allows the storage of over 1000 recipes to be stored, and a USB port enables upgrades and downloads of log files.
To find out more about the Q Plus Series of coaters, view our brochure or get in touch with us today.
To view our latest webinars on coating technologies, we invite you to view our series here:
- Practical advice in sample preparation for SEM
- How to achieve appropriate metal coating quality for my application
- Carbon coatings and Glow Discharge for TEM
References and Further Reading
- The Diffraction Barrier in Optical Microscopy. Nikon’s MicroscopyU https://www.microscopyu.com/techniques/super-resolution/the-diffraction-barrier-in-optical-microscopy.
- Goldstein, J. I. et al. Coating Techniques for SEM and Microanalysis. in Scanning Electron Microscopy and X-Ray Microanalysis: A Text for Biologist, Materials Scientist, and Geologists (eds. Goldstein, J. I. et al.) 461–494 (Springer US, 1981). doi:10.1007/978-1-4613-3273-2_10.
- Ahire, J. J., Neveling, D. P. & Dicks, L. M. T. Polyacrylonitrile (PAN) nanofibres spun with copper nanoparticles: an anti-Escherichia coli membrane for water treatment. Appl Microbiol Biotechnol 102, 7171–7181 (2018).